This fun and super cool Fizzy Frozen Eggs Easter Science Experiment introduces chemical reactions in a very festive manner. Use this in the classroom, homeschool, or anytime you want to show your kids something cool!

Hands on activities are always a great way for kids to learn. If they can SEE it and BE a part of it, they are more likely to remember things. This Easter science experiment shows kids how chemical reactions work. Sometimes, when two substances are combined a completely new substance is created.
For this experiment, when you mix the baking soda (base) with vinegar (acid) it results in carbon dioxide being created and released. That’s what you see when the eggs start fizzing!
Before you combine the baking soda eggs and vinegar, have the kids predict what they think will happen.
What You’ll Need for this Fizzy Frozen Eggs Experiment
- ¾ cup baking soda
- ¼ cup warm water
- Food coloring
- Vinegar
- Egg-shape silicone mold
- Shallow bowl

In a large mixing cup, stir together the baking soda and water until the baking soda is dissolved. Stir in food coloring. We used pastel colors for our Easter eggs but any color will work!

Pour the mixture into the egg-shaped silicone mold and freeze overnight or until completely frozen.
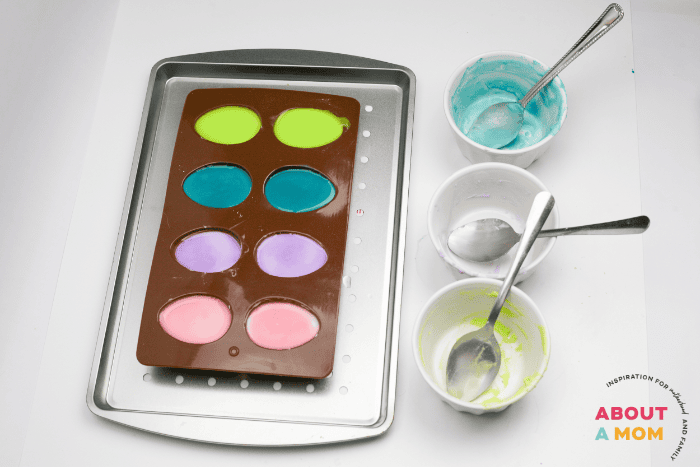
Carefully remove frozen eggs from the mold.

Pour vinegar into the shallow bowl.
Carefully lower the frozen egg into the vinegar. You can also coat the eggs in vinegar using a syringe. Either way, you will notice the fizzing immediately!

Alternative experiment ideas:
You can mix up your Easter science experiment to test new reactions and theories!
- Create frozen eggs using a solution made from vinegar and water. Dip the frozen eggs into a solution of baking soda and water.
- Experiment with different ratios of baking soda to water when freezing the eggs to see which brings a more exciting reaction.

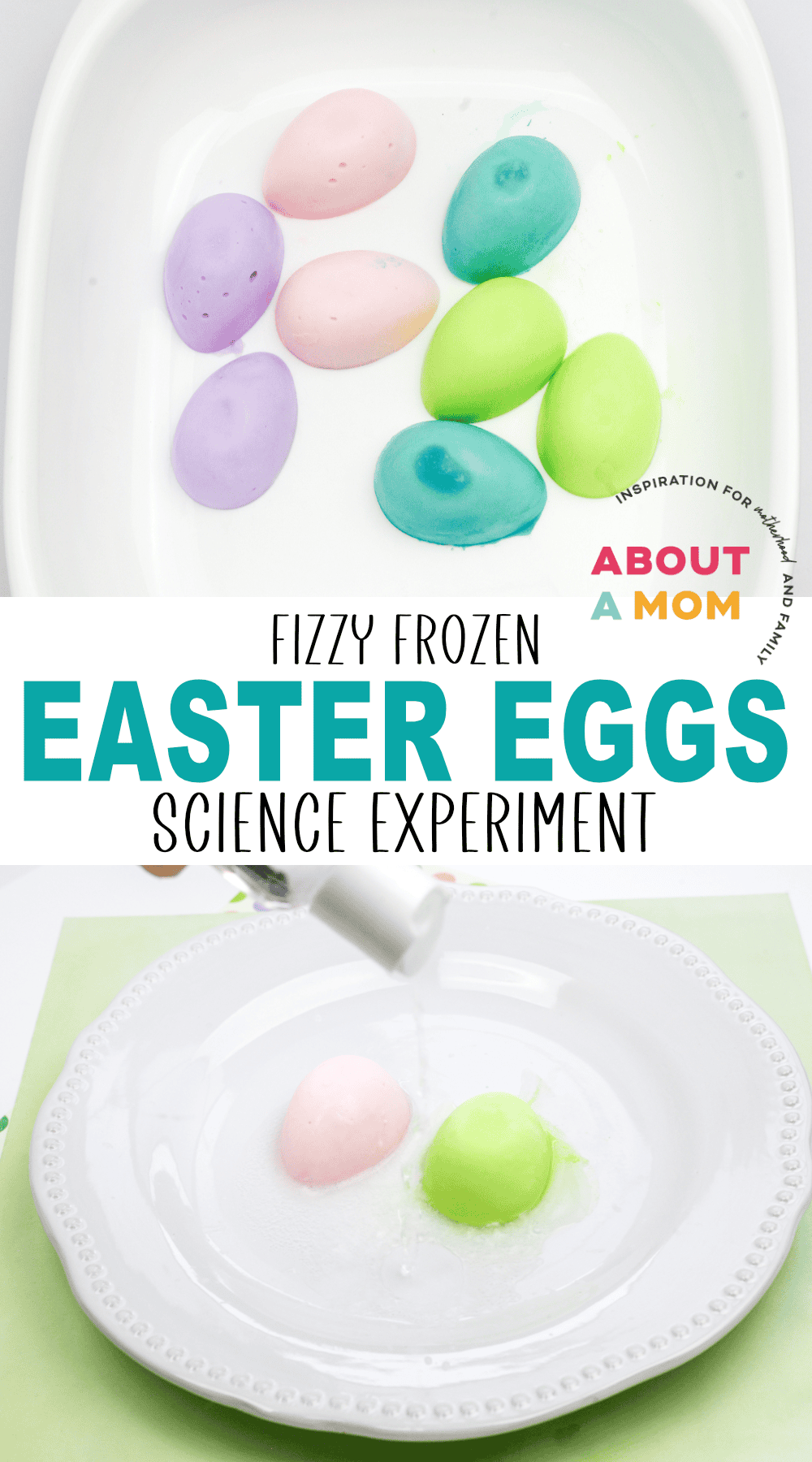
Leave a Comment